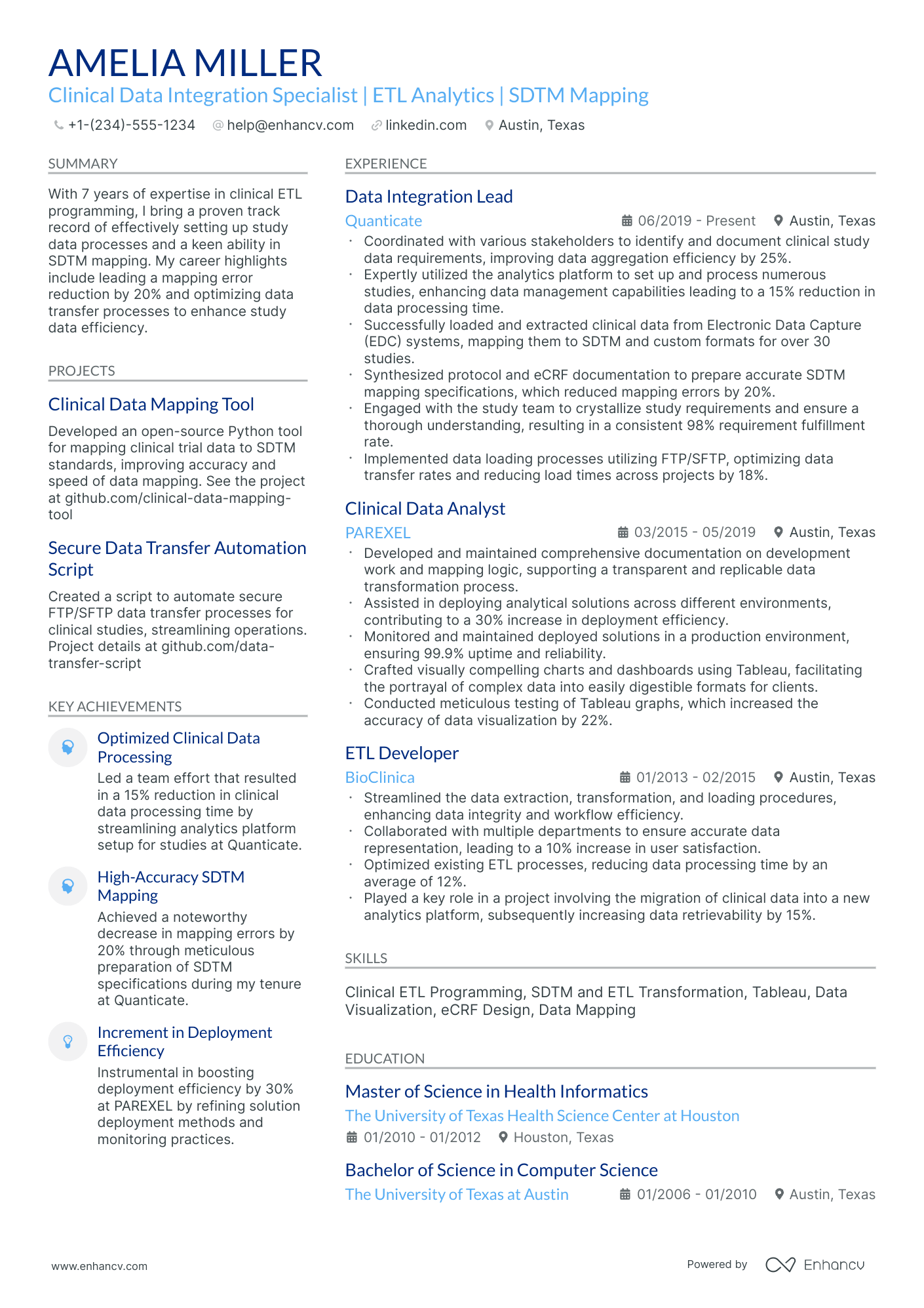
Many SAS clinical programmer resumes fail because they read like task logs and bury validation, traceability, and submission-ready evidence. This SAS clinical programmer resume guide helps you pass ATS filters, recruiter skims, and intense competition. If you're unsure where to start, understanding how to write a resume that highlights outcomes rather than duties is the first step.
A strong resume shows outcomes: how you improved data quality, shortened delivery timelines, and reduced rework. You should highlight SDTM and ADaM build scope, audit findings avoided, programming cycle time cut, tables and listings delivered, and reviewer questions resolved.
Key takeaways
- Quantify clinical programming outcomes like cycle time, error reduction, and submission milestones in every bullet.
- Mirror exact SAS tools, CDISC standards, and trial phases from the job posting in your experience section.
- Use reverse-chronological format for senior roles and hybrid format for career switchers or junior candidates.
- Pair every listed skill with a specific action and measurable result to prove applied competence.
- Include SDTM, ADaM, and TLF deliverables prominently—recruiters and ATS filters prioritize these terms.
- Build entry-level credibility through certification projects, coursework, and a GitHub portfolio of SAS code.
- Use Enhancv to turn routine programming tasks into recruiter-ready, metric-driven resume bullets faster.
How to format a SAS clinical programmer resume
Recruiters hiring for SAS clinical programmer roles prioritize demonstrated proficiency in SAS programming (Base SAS, SAS/STAT, SAS macros), familiarity with CDISC standards (SDTM, ADaM), and hands-on experience generating tables, listings, and figures (TLFs) for regulatory submissions. A clean, well-structured resume format ensures these technical competencies and your clinical trial experience surface quickly during both automated screening and manual review.
I have significant experience in this role—which format should I use?
Use the reverse-chronological format—it's the strongest choice for showcasing progressive SAS programming experience across clinical studies and therapeutic areas. Do:
- Lead each role entry with your scope: number of studies supported, phases (I–IV), team size, and regulatory submission involvement.
- Highlight role-specific tools and standards front and center—SAS Base, SAS/STAT, SAS macros, CDISC SDTM/ADaM, PROC SQL, ODS, and platforms like Pinnacle 21 or Define.xml.
- Quantify outcomes tied to timelines, data quality, or submission readiness rather than listing routine tasks.
I'm junior or switching into this role—what format works best?
A hybrid format works best because it lets you lead with a dedicated technical skills section while still providing a chronological work history that shows how you've applied those skills. Do:
- Place a prominent skills section near the top listing SAS programming competencies (Base SAS, SAS macros, PROC SQL, ODS), CDISC standards, and any relevant statistical or clinical data management tools.
- Feature academic projects, internships, or transitional experience—such as biostatistics coursework, SAS certification projects, or data analysis work in adjacent fields—as concrete evidence of capability.
- Connect every listed skill to a specific action and a measurable or observable result to demonstrate applied competence.
Why not use a functional resume?
A functional format strips away the clinical trial context—study phases, therapeutic areas, regulatory timelines—that hiring managers rely on to assess whether your SAS programming skills have been tested in real submission environments, making it significantly harder to evaluate your readiness for the role. A functional format may be acceptable if you're transitioning from a non-clinical SAS role (such as SAS work in finance or marketing analytics) or re-entering the workforce after a gap, but only if you tie every listed skill directly to specific projects, datasets, or measurable outcomes rather than presenting skills in isolation.
With your resume's structure and layout in place, the next step is determining which sections to include so each one reinforces your qualifications as a SAS clinical programmer.
What sections should go on a SAS clinical programmer resume
Recruiters expect a clean, compliance-focused resume that proves you can deliver validated clinical programming outputs in regulated environments. Knowing which resume sections to include—and in what order—ensures nothing critical gets overlooked.
Use this structure for maximum clarity:
- Header
- Summary
- Experience
- Skills
- Projects
- Education
- Certifications
- Optional sections: Publications, Research, Languages
Strong experience bullets should emphasize the clinical deliverables you produced, the validation and compliance standards you met, and the measurable impact on timelines, data quality, and submission readiness.
Is your resume good enough?
Drop your resume here or choose a file. PDF & DOCX only. Max 2MB file size.
Once you’ve organized your resume with the right core components, the next step is to write your SAS clinical programmer resume experience so each entry supports that structure with clear, role-specific impact.
How to write your SAS clinical programmer resume experience
Your work experience section should spotlight the clinical programming work you've shipped—CDSC/ADaM datasets delivered, SDTM mappings validated, regulatory submissions supported—using the specific SAS tools and statistical methods that drove each outcome. Hiring managers prioritize demonstrated impact on data integrity, submission timelines, and regulatory compliance over descriptive task lists of routine programming duties.
Each entry should include:
- Job title
- Company and location (or remote)
- Dates of employment (month and year)
Three to five concise bullet points showing what you owned, how you executed, and what outcomes you delivered:
- Ownership scope: the clinical studies, therapeutic areas, datasets, submission packages, or programming teams you were directly accountable for as a SAS clinical programmer.
- Execution approach: the SAS procedures, macros, CDISC standards, data integration frameworks, or validation methods you applied to build, test, and deliver analysis-ready datasets and outputs.
- Value improved: the changes you drove in data quality, programming efficiency, submission readiness, regulatory compliance, or error reduction across your clinical programming deliverables.
- Collaboration context: how you partnered with biostatisticians, data managers, medical writers, regulatory affairs teams, or external CRO partners to align programming outputs with study protocols and reporting requirements.
- Impact delivered: the outcomes your work produced—expressed through submission milestones met, audit findings resolved, review cycles shortened, or portfolio-level contributions—rather than a summary of daily programming activity.
Experience bullet formula
A SAS clinical programmer experience example
✅ Right example - modern, quantified, specific.
SAS Clinical Programmer II
Apex Clinical Research | Remote
2022–Present
Mid-size contract research organization supporting Phase II–III oncology and immunology trials for global sponsors.
- Programmed Study Data Tabulation Model (SDTM) and Analysis Data Model (ADaM) datasets in SAS 9.4 using Pinnacle 21 validation, cutting validation findings by 38% across three studies.
- Automated Tables, Listings, and Figures (TLFs) with SAS macros and ODS RTF, reducing production cycle time from eight days to five days per interim analysis.
- Developed Define-XML and annotated Case Report Form (CRF) traceability with SAS Clinical Standards Toolkit, achieving zero critical issues in two consecutive sponsor audits.
- Reconciled clinical and safety data with MedDRA and WHO Drug dictionaries and Data Clarification Forms (DCFs), lowering query rate by 22% and improving database lock readiness by two weeks.
- Partnered with biostatisticians, data managers, and clinical operations to implement controlled terminology updates and derivation specifications, preventing three protocol deviation-related rework cycles.
Now that you've seen what a strong experience section looks like in practice, let's break down how to customize yours to match the specific job you're targeting.
How to tailor your SAS clinical programmer resume experience
Recruiters evaluate your SAS clinical programmer resume through both human review and applicant tracking systems. Tailoring your resume to the job description ensures your qualifications connect directly with what hiring teams prioritize.
Ways to tailor your SAS clinical programmer experience:
- Match SAS procedures and macros specifically referenced in the job posting.
- Mirror the exact clinical trial phases the employer highlights.
- Align your CDISC standards experience with SDTM or ADaM requirements listed.
- Reflect the statistical reporting tools or outputs the role requires.
- Incorporate regulatory submission experience when the posting mentions FDA or EMA.
- Use the same terminology for SOPs and programming workflows described.
- Highlight therapeutic area knowledge that matches the employer's pipeline focus.
- Emphasize quality control and validation processes the job description prioritizes.
Tailoring means aligning your real accomplishments with the role's stated requirements, not forcing keywords where they don't belong.
Resume tailoring examples for SAS clinical programmer
| Job description excerpt | Untailored | Tailored |
|---|---|---|
| Develop and validate SAS programs for SDTM and ADaM datasets in compliance with CDISC standards for Phase III oncology trials. | Worked with SAS to create datasets for clinical studies. | Developed and validated 50+ SAS programs generating SDTM and ADaM datasets under CDISC standards for two Phase III oncology trials, passing all FDA submission audits with zero critical findings. |
| Generate tables, listings, and figures (TLFs) using SAS/BASE, SAS/STAT, and SAS/GRAPH to support integrated summary of safety (ISS) and efficacy (ISE) submissions. | Created reports and tables for clinical teams using SAS. | Produced 120+ tables, listings, and figures using SAS/BASE, SAS/STAT, and SAS/GRAPH to support ISS and ISE submissions, reducing QC cycle time by 30% through reusable macro libraries. |
| Perform data integrity checks and write SAS macros to automate SDTM mapping across multiple therapeutic areas, including cardiovascular and immunology. | Helped with data checks and some automation tasks in SAS. | Built and maintained 15 SAS macros automating SDTM mapping and data integrity checks across four concurrent cardiovascular and immunology studies, cutting manual mapping effort by 40%. |
Once you’ve aligned your experience with the role’s clinical programming needs, the next step is to quantify your SAS clinical programmer achievements so hiring teams can quickly see your impact.
How to quantify your SAS clinical programmer achievements
Quantifying your achievements proves you improve study delivery, data quality, and regulatory readiness. Use numbers tied to cycle time, validation defects, SDTM and ADaM compliance, rework avoided, and volume of outputs delivered across studies.
Quantifying examples for SAS clinical programmer
| Metric | Example |
|---|---|
| Delivery speed | "Cut TLF delivery from twelve to seven business days by automating SAS macros and standardizing shells across three Phase III studies." |
| Data quality | "Reduced SDTM validation errors by 45% by resolving Pinnacle 21 findings and adding SAS checks for controlled terminology and key variables." |
| Throughput | "Generated 180+ TLFs and fifteen ADaM datasets per interim analysis using SAS and Git, meeting all four biweekly data cut deadlines." |
| Compliance readiness | "Achieved zero critical FDA submission findings by producing define.xml, reviewer's guides, and traceability from SDTM to ADaM to TLFs." |
| Rework reduction | "Lowered post-QC rework by 30% by implementing double-programming standards and automated compare outputs in SAS for key endpoints." |
Turn your everyday tasks into measurable, recruiter-ready resume bullets in seconds with Enhancv's Bullet Point Generator.
Once you've crafted strong bullet points for your experience section, it's equally important to strategically present your hard and soft skills to give hiring managers a complete picture of your qualifications.
How to list your hard and soft skills on a SAS clinical programmer resume
Your skills section shows you can deliver compliant clinical datasets and outputs, and recruiters and an ATS (applicant tracking system) scan this section to confirm tool and domain fit, so aim for a hard-skill-heavy mix with targeted collaboration and soft skills.
SAS clinical programmer roles require a blend of:
- Product strategy and discovery skills.
- Data, analytics, and experimentation skills.
- Delivery, execution, and go-to-market discipline.
- Soft skills.
Your skills section should be:
- Scannable (bullet-style grouping).
- Relevant to the job post.
- Backed by proof in experience bullets.
- Updated with current tools.
Place your skills section:
- Above experience if you're junior or switching careers.
- Below experience if you're mid/senior with strong achievements.
Hard skills
- Base SAS, SAS Macro
- SAS Clinical, SAS/STAT
- CDISC SDTM, ADaM
- Define-XML, Controlled Terminology
- TLF programming, QC
- Pinnacle 21 validation
- MedDRA, WHODrug coding support
- Oracle Clinical, Medidata Rave
- Clinical data review listings
- SQL, PROC SQL
- Git version control
- 21 CFR Part 11 compliance
Soft skills
- Translate SAP to specs
- Clarify ambiguous requirements
- Partner with biostatistics and data management
- Document assumptions and decisions
- Prioritize programming deliverables
- Estimate effort and timelines
- Communicate status and risks early
- Resolve validation findings fast
- Maintain audit-ready traceability
- Review code with peer feedback
- Coordinate across global teams
- Own quality through double-programming checks
How to show your SAS clinical programmer skills in context
Skills shouldn't live only in a bulleted list on your resume. Explore resume skills examples to see how top candidates integrate competencies throughout their documents.
They should be demonstrated in:
- Your summary (high-level professional identity)
- Your experience (proof through outcomes)
Here's what that looks like in practice.
Summary example
Senior SAS clinical programmer with 10 years in oncology trials. Built automated SDTM/ADaM workflows that cut dataset delivery timelines by 35%. Skilled in CDISC standards, regulatory submissions, and cross-functional collaboration with biostatistics and data management teams.
- Reflects senior-level expertise immediately
- Names industry-standard tools and frameworks
- Quantifies impact with a clear metric
- Highlights collaboration as a soft skill
Experience example
Senior SAS Clinical Programmer
Parexel | Remote
June 2019–March 2024
- Developed and validated 120+ SDTM and ADaM datasets across four Phase III oncology trials using SAS 9.4 and Base SAS macros.
- Collaborated with biostatisticians and medical writers to produce TLFs, reducing review cycles by 25% through standardized macro libraries.
- Led CDISC compliance audits for two FDA submissions, resolving 98% of define.xml discrepancies before the first regulatory review.
- Every bullet includes a measurable outcome.
- Tools and collaboration appear naturally in context.
Once you’ve demonstrated your SAS clinical programmer capabilities through relevant projects and results, the next step is applying that same approach to build a strong SAS clinical programmer resume even without formal experience.
How do I write a SAS clinical programmer resume with no experience
Even without full-time experience, you can demonstrate readiness through building a resume without work experience that leverages projects, coursework, and certifications:
- Clinical trial datasets course projects
- SAS Base certification practice work
- CDISC SDTM mapping assignments
- ADaM derivations in capstones
- Mock TLFs using PROC REPORT
- Data cleaning with SAS macros
- GitHub portfolio of SAS code
- Volunteer data work for researchers
Focus on:
- SDTM and ADaM deliverables
- SAS code quality and validation
- TLF production and QC
- Clear documentation and traceability
Resume format tip for entry-level SAS clinical programmer
Use a combination resume format to lead with projects, tools, and deliverables, then list education and any relevant work history. Do:
- Put SDTM, ADaM, and TLFs first.
- Add a Projects section with metrics.
- Name tools: SAS, SQL, Git.
- Show QC methods and log checks.
- Link to a GitHub code sample.
- Built SDTM DM and AE from raw CSVs in SAS, applied controlled terminology, and reduced validation warnings by 40% through iterative log review and QC.
When hands-on experience is limited, your education section becomes one of the strongest tools for demonstrating relevant knowledge and qualifications.
How to list your education on a SAS clinical programmer resume
Your education section helps hiring teams confirm you have the technical and analytical foundation needed. It validates your readiness for SAS clinical programmer responsibilities in regulated environments.
Include:
- Degree name
- Institution
- Location
- Graduation year
- Relevant coursework (for juniors or entry-level candidates)
- Honors & GPA (if 3.5 or higher)
Skip month and day details—list only the graduation year.
Here's a strong education entry tailored for a SAS clinical programmer resume:
Example education entry
Bachelor of Science in Biostatistics
University of North Carolina at Chapel Hill, Chapel Hill, NC
Graduated: 2021
GPA: 3.7/4.0
- Relevant Coursework: Clinical Trial Design, SAS Programming for Health Sciences, Survival Analysis, Statistical Methods in Epidemiology
- Honors: Dean's List (six semesters), Phi Beta Kappa Honor Society
How to list your certifications on a SAS clinical programmer resume
Certifications on your resume show your commitment to learning, confirm tool proficiency, and prove industry relevance as a SAS clinical programmer. They also help recruiters validate your skills quickly.
Include:
- Certificate name
- Issuing organization
- Year
- Optional: credential ID or URL
- Place certifications below education if they're older, less relevant, or you have a strong, recent degree for a SAS clinical programmer role.
- Place certifications above education if they're recent, highly relevant, or required for the target SAS clinical programmer position.
Best certifications for your SAS clinical programmer resume
- SAS Certified Specialist: Base Programming Using SAS 9.4
- SAS Certified Professional: Advanced Programming Using SAS 9.4
- SAS Certified Clinical Trials Programmer Using SAS 9
- Certified Clinical Research Associate (CCRA)
- Certified Clinical Research Professional (CCRP)
- CDISC Certified Professional (CDISC-CP)
- Good Clinical Practice (GCP) Certification
Once you’ve positioned your credentials to validate your SAS clinical programming expertise, shift to your resume summary to highlight that value upfront.
How to write your SAS clinical programmer resume summary
Your resume summary is the first thing a recruiter reads, so it must immediately signal your fit for the role. A strong opening positions you as a qualified SAS clinical programmer before the hiring manager scrolls any further.
Keep it to three to four lines, with:
- Your title and total years of SAS clinical programming experience.
- The therapeutic areas or clinical trial phases you've worked in.
- Core tools like SAS Base, SAS/STAT, CDISC standards, SDTM, and ADaM.
- One or two measurable results, such as timelines met or submissions supported.
- Collaboration and attention to detail demonstrated through real project outcomes.
PRO TIP
At the entry or mid level, focus on specific SAS skills, CDISC knowledge, and early contributions to clinical studies. Avoid vague claims like "passionate team player" or "quick learner." Instead, name the tools you've used and the outputs you've delivered. Recruiters want proof of relevance, not motivation.
Example summary for a SAS clinical programmer
SAS clinical programmer with three years of experience developing SDTM and ADaM datasets for Phase II–III oncology trials. Delivered submission-ready outputs for two successful FDA filings using SAS Base and SAS/STAT.
Optimize your resume summary and objective for ATS
Drop your resume here or choose a file.
PDF & DOCX only. Max 2MB file size.
Now that your summary captures your strongest qualifications, make sure your header presents the essential contact and professional details recruiters need to reach you.
What to include in a SAS clinical programmer resume header
Your resume header lists your key identity and contact details, helping SAS clinical programmer candidates boost visibility, credibility, and recruiter screening speed.
Essential resume header elements
- Full name
- Tailored job title and headline
- Location
- Phone number
- Professional email
- GitHub link
- Portfolio link
A LinkedIn link helps recruiters verify your experience quickly and supports screening.
Don't include photos on a SAS clinical programmer resume unless the role is explicitly front-facing or appearance-dependent.
Use a clear SAS clinical programmer title, keep links readable, and match your header details to your application and LinkedIn profile.
Example
SAS clinical programmer resume header
Jordan Taylor
SAS Clinical Programmer | SDTM and ADaM Programming | Clinical Trial Reporting
Boston, MA
(617) 555-01XX
jordan.taylor@enhancv.com
github.com/jordantaylor
jordantaylor.com
linkedin.com/in/jordantaylor
Once your contact details and role identifiers are set at the top, add targeted additional sections to reinforce your qualifications and make the resume more complete.
Additional sections for SAS clinical programmer resumes
Adding extra sections helps you stand out when your core qualifications match other candidates and you need stronger differentiation.
Choose sections that reinforce your clinical programming expertise, regulatory knowledge, or industry involvement:
- Certifications (SAS Base, SAS Advanced, SAS Clinical Trials)
- Publications and conference presentations
- Professional affiliations (PharmaSUG, CDISC, DIA)
- Languages
- Continuing education and specialized training
- Hobbies and interests
- Volunteer experience in healthcare or data science
Once you've strengthened your resume with targeted additional sections, the next step is pairing it with a well-crafted cover letter to make an even stronger impression.
Do SAS clinical programmer resumes need a cover letter
A cover letter isn't required for a SAS clinical programmer, but it helps in competitive roles or when hiring teams expect one. It can make a difference when your resume needs context or you want to show clear fit.
Use a cover letter to add details your resume can't:
- Explain role and team fit by matching your experience to the trial phase, therapeutic area, and standards the group uses.
- Highlight one or two relevant projects or outcomes, such as faster table, listing, and figure delivery or cleaner ADaM and SDTM datasets.
- Show understanding of the product, users, or business context, like how outputs support clinical review, submissions, and audit readiness.
- Address career transitions or non-obvious experience by linking transferable skills to SAS clinical programmer work and regulated workflows.
Drop your resume here or choose a file.
PDF & DOCX only. Max 2MB file size.
Even without relying on a cover letter to add context, using AI to improve your SAS clinical programmer resume helps you sharpen the document itself and align it more closely with the role.
Using AI to improve your SAS clinical programmer resume
AI can sharpen your resume's clarity, structure, and overall impact. It helps you find stronger phrasing and spot weak spots fast. But overuse kills authenticity. Once your content feels clear and role-aligned, step away from AI assistance. For specific prompt ideas, check out how ChatGPT can help with resume writing.
Here are 10 practical prompts to strengthen specific sections of your SAS clinical programmer resume:
Sharpen your summary
Quantify experience bullets
Align skills section
Strengthen action verbs
Tailor to job posting
Clarify project descriptions
Refine education details
Highlight certifications
Eliminate redundancy
Improve CDISC references
Conclusion
A strong SAS clinical programmer resume proves impact with measurable outcomes, role-specific skills, and a clear structure. It highlights study support results, programming accuracy, and delivery speed, backed by metrics. It stays easy to scan with consistent headings and targeted bullets.
This approach shows you can meet today’s expectations and adapt to near-future hiring needs. It signals you can deliver reliable outputs, follow standards, and collaborate across clinical teams. Keep it focused, quantified, and structured, and you’ll compete well.