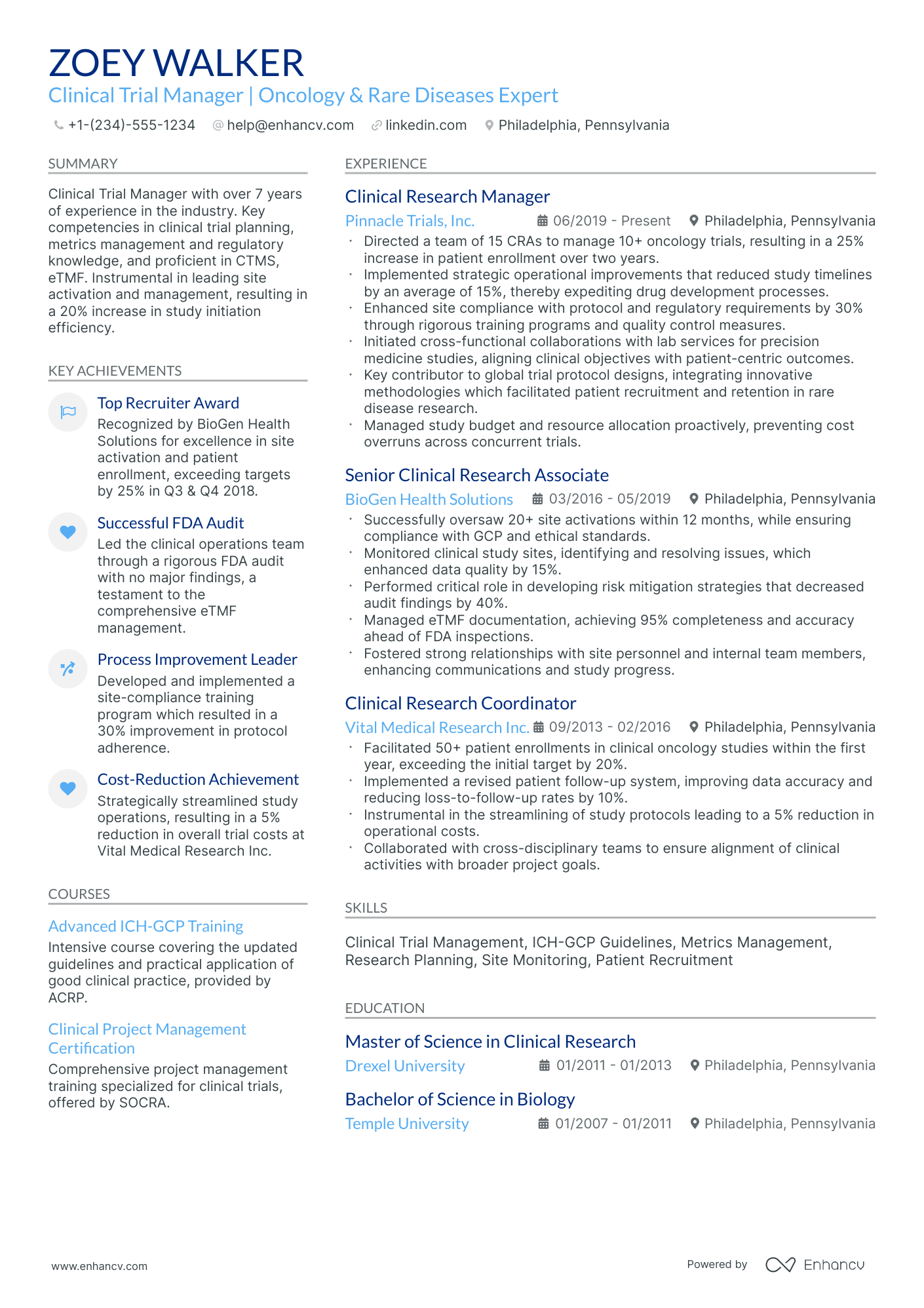
Clinical trial manager resume drafts often fail because they read like task logs and bury evidence of oversight, compliance, and delivery. That hurts in ATS screening and rapid recruiter scans, where competition is high and proof must surface fast.
A strong resume shows what you delivered and how you reduced risk. Knowing how to make your resume stand out means highlighting enrollment gains, on-time milestones across countries, protocol deviations reduced, audit readiness, query cycle time, database lock speed, and budget variance.
Key takeaways
- Quantify enrollment speed, protocol deviations, and budget variance to prove trial delivery impact.
- Use reverse-chronological format for senior roles to show expanding oversight clearly.
- Tailor experience bullets to mirror each job posting's tools, phases, and compliance language.
- Place skills above experience if you're junior or transitioning into clinical trial management.
- Pair every listed skill with measurable proof inside your experience section.
- Certifications like ACRP-CP or SOCRA-CCRP belong near education to reinforce specialized credibility.
- Use Enhancv to turn routine trial tasks into results-focused, recruiter-ready resume bullets.
Job market snapshot for clinical trial managers
We analyzed 106 recent clinical trial manager job ads across major US job boards. These numbers help you understand employer expectations, top companies hiring, salary landscape at a glance.
What level of experience employers are looking for clinical trial managers
| Years of Experience | Percentage found in job ads |
|---|---|
| 1–2 years | 8.5% (9) |
| 3–4 years | 28.3% (30) |
| 5–6 years | 22.6% (24) |
| 7–8 years | 10.4% (11) |
| 9–10 years | 0.9% (1) |
| 10+ years | 0.9% (1) |
| Not specified | 29.2% (31) |
Clinical trial manager ads by area of specialization (industry)
| Industry (Area) | Percentage found in job ads |
|---|---|
| Healthcare | 57.5% (61) |
| Finance & Banking | 39.6% (42) |
Top companies hiring clinical trial managers
| Company | Percentage found in job ads |
|---|---|
| Parexel | 20.8% (22) |
| Syneos Health, Inc | 16.0% (17) |
Role overview stats
These tables show the most common responsibilities and employment types for clinical trial manager roles. Use them to align your resume with what employers expect and to understand how the role is structured across the market.
Day-to-day activities and top responsibilities for a clinical trial manager
| Responsibility | Percentage found in job ads |
|---|---|
| Clinical trial management | 61.3% (65) |
| Vendor management | 39.6% (42) |
| Clinical operations | 33.0% (35) |
| Clinical monitoring | 24.5% (26) |
| Clinical research | 22.6% (24) |
| Gcp | 22.6% (24) |
| Oncology | 17.0% (18) |
| Query resolution | 17.0% (18) |
| Good clinical practice | 16.0% (17) |
| Project management | 16.0% (17) |
| Fda regulations | 15.1% (16) |
| Audit support | 13.2% (14) |
Type of employment (remote vs on-site vs hybrid)
| Employment type | Percentage found in job ads |
|---|---|
| Remote | 55.7% (59) |
| On-site | 22.6% (24) |
| Hybrid | 21.7% (23) |
How to format a clinical trial manager resume
Recruiters evaluating clinical trial manager candidates prioritize evidence of trial oversight from protocol development through closeout, cross-functional team leadership, regulatory compliance accountability, and measurable impact on timelines and budgets. A reverse-chronological format surfaces these signals immediately by presenting your career progression and expanding scope in the order hiring managers expect to see it.
I have significant experience in this role—which format should I use?
Use a reverse-chronological format—it's the strongest choice for showcasing your depth of clinical trial management experience and growing oversight responsibilities. Do:
- Lead with your most recent role and clearly define the scope of trials you managed, including phases, therapeutic areas, number of sites, and team size.
- Highlight role-specific expertise such as CTMS and EDC platforms (e.g., Medidata Rave, Oracle InForm, Veeva Vault), GCP/ICH compliance, CRO management, and regulatory submission support.
- Quantify outcomes tied to enrollment targets, budget adherence, audit results, and timeline performance.
I'm junior or switching into this role—what format works best?
A hybrid format works best because it lets you lead with relevant clinical research skills while still providing a concise work history that shows progression. Do:
- Place a dedicated skills section near the top that highlights competencies such as GCP knowledge, site monitoring, protocol compliance, CTMS tools, and regulatory documentation.
- Feature project-based experience—such as CRA rotations, clinical research coordinator work, or cross-functional study support—even if your formal title wasn't "clinical trial manager."
- Connect every action to a measurable or observable result so recruiters can see your direct contribution.
Why hybrid and functional resumes don't work for senior roles
Hybrid formats push your leadership timeline below a skills summary, making it harder for recruiters to trace how your trial oversight, budget authority, and regulatory accountability expanded over time. Functional formats strip away role context entirely, diluting evidence of decision ownership across complex, multi-site programs and obscuring the progression from site-level management to strategic portfolio oversight. Choosing the right resume layout is critical to presenting your career trajectory clearly. Avoid both formats if you have five or more years of progressive clinical trial management experience—they'll weaken your candidacy rather than strengthen it.
- Edge-case exception: A functional format may be acceptable only if you're transitioning into clinical trial management from an adjacent role (e.g., CRA, regulatory affairs, or clinical operations) and lack direct management titles, but you must still tie every listed skill to specific trial projects, measurable outcomes, and compliance results.
Once your format establishes a clean, scannable structure, you'll need to fill it with the right sections to give recruiters exactly what they're looking for.
What sections should go on a clinical trial manager resume
Recruiters expect a clinical trial manager resume to show end-to-end trial oversight, compliance, and measurable delivery across timelines, budgets, and quality. Understanding which resume sections to include ensures you present a complete picture of your qualifications.
Use this structure for maximum clarity:
- Header
- Summary
- Experience
- Skills
- Projects
- Education
- Certifications
- Optional sections: Publications, Leadership, Languages
Your experience bullets should emphasize trial phase and therapeutic area scope, enrollment and timeline outcomes, budget and vendor oversight, inspection readiness, and quantifiable quality and compliance results.
Is your resume good enough?
Drop your resume here or choose a file. PDF & DOCX only. Max 2MB file size.
Once you’ve organized your resume with the right core components, the next step is to write your clinical trial manager resume experience section so it aligns with that structure and shows your impact clearly.
How to write your clinical trial manager resume experience
The experience section is where you prove you've delivered real results—managing trial timelines, overseeing regulatory submissions, coordinating site operations, and driving study milestones using role-specific tools and methodologies. Hiring managers prioritize demonstrated impact over descriptive task lists, so every bullet should connect what you owned to a measurable outcome.
Each entry should include:
- Job title
- Company and location (or remote)
- Dates of employment (month and year)
Three to five concise bullet points showing what you owned, how you executed, and what outcomes you delivered:
- Ownership scope: the clinical trials, therapeutic areas, study phases, site networks, or cross-functional study teams you were directly accountable for as a clinical trial manager.
- Execution approach: the tools, frameworks, and methods you relied on to drive decisions and deliver work—such as clinical trial management systems, electronic data capture platforms, risk-based monitoring strategies, or regulatory submission processes.
- Value improved: the specific dimensions of trial performance you strengthened, whether that involved enrollment timelines, protocol compliance, data quality, patient safety reporting, site activation speed, or audit readiness.
- Collaboration context: how you partnered with sponsors, contract research organizations, principal investigators, regulatory authorities, data management teams, or internal medical affairs and pharmacovigilance stakeholders to advance study objectives.
- Impact delivered: the tangible outcomes your work produced, framed through results, scale, or business impact—such as trials brought to completion, regulatory milestones met, deviations reduced, or portfolio timelines accelerated—rather than a list of activities performed.
Experience bullet formula
A clinical trial manager experience example
✅ Right example - modern, quantified, specific.
Clinical Trial Manager
Arcadia Biopharma | Boston, MA
2021–Present
Clinical-stage biotech running global Phase II–III oncology trials across North America and Europe.
- Led end-to-end execution of three Phase II oncology trials (twenty-one sites, four hundred twelve participants), delivering last patient in four weeks ahead of plan by tightening the critical path in Microsoft Project and aligning CRO (contract research organization) workstreams.
- Implemented risk-based monitoring and centralized monitoring dashboards in Medidata Rave and Power BI, cutting protocol deviations by 28% and reducing on-site monitoring days by 22% while maintaining data quality thresholds.
- Drove EDC (electronic data capture) build, UAT (user acceptance testing), and mid-study amendments with clinical data management, biostatistics, and vendors, reducing query cycle time from nine days to five days and improving database lock readiness by 18%.
- Negotiated and managed CRO and central lab performance against KPIs (key performance indicators) using CTMS (clinical trial management system) and vendor scorecards, improving on-time visit window compliance from 84% to 94% and avoiding $350K in projected change orders.
- Coordinated cross-functional governance with clinical operations, pharmacovigilance, medical monitors, and regulatory affairs, achieving zero critical audit findings across two sponsor audits and one GCP (Good Clinical Practice) inspection readiness assessment.
Now that you've seen what a strong experience section looks like in practice, let's break down how to customize yours for each specific job posting.
How to tailor your clinical trial manager resume experience
Recruiters evaluate your clinical trial manager resume through both applicant tracking systems and manual review. Tailoring your resume to the job description ensures your qualifications stand out in both screening methods.
Ways to tailor your clinical trial manager experience:
- Match clinical trial management systems and EDC platforms named in the posting.
- Mirror the exact regulatory standards like ICH-GCP or FDA guidelines listed.
- Reflect site monitoring visit types and frequencies the role specifies.
- Use the same terminology for study phases referenced in the description.
- Highlight therapeutic area experience when the posting names specific indications.
- Emphasize cross-functional collaboration with CROs or sponsors if mentioned.
- Align your quality oversight language with compliance frameworks they reference.
- Include protocol development or amendment processes the role prioritizes.
Tailoring means aligning your real accomplishments with the employer's stated requirements—not forcing keywords where they don't belong.
Resume tailoring examples for clinical trial manager
| Job description excerpt | Untailored | Tailored |
|---|---|---|
| Oversee Phase II–III oncology trials using Medidata Rave, ensuring compliance with ICH-GCP and FDA 21 CFR Part 11 regulations | Managed clinical trials and ensured regulatory compliance across multiple projects. | Directed four Phase II–III oncology trials enrolling 1,200+ patients, managing end-to-end data collection in Medidata Rave while maintaining full compliance with ICH-GCP and FDA 21 CFR Part 11 requirements. |
| Coordinate cross-functional teams across 15+ global sites, managing CROs and driving enrollment targets using CTMS (Veeva Vault) | Worked with internal and external teams to support clinical trial operations. | Coordinated cross-functional teams across 18 global sites in three countries, managing two CROs and tracking enrollment milestones in Veeva Vault CTMS to exceed patient recruitment targets by 12%. |
| Develop and maintain risk-based monitoring plans, conduct root cause analyses for protocol deviations, and present findings to sponsor stakeholders | Helped identify issues during trials and communicated updates to leadership. | Built and executed risk-based monitoring plans for five concurrent studies, led root cause analyses that reduced protocol deviations by 30%, and delivered quarterly findings presentations to sponsor stakeholders. |
Once you’ve aligned your experience with the role’s needs, the next step is to quantify your clinical trial manager achievements so hiring teams can see the measurable impact of your work.
How to quantify your clinical trial manager achievements
Quantifying your achievements shows how you improved timelines, compliance, quality, and budget across studies. Focus on enrollment speed, protocol deviation rates, monitoring cycle time, audit findings, site activation timelines, and cost per patient.
Quantifying examples for clinical trial manager
| Metric | Example |
|---|---|
| Enrollment speed | "Accelerated enrollment by 18% across eight sites by tightening referral workflows and tracking weekly KPIs in Microsoft Excel and Power BI." |
| Compliance quality | "Reduced major protocol deviations from 2.6% to 1.1% by retraining sites, revising checklists, and enforcing ICH E6 documentation standards." |
| Site activation time | "Cut site activation median time from 12 weeks to eight weeks by standardizing feasibility, budget templates, and ethics submission packages." |
| Monitoring efficiency | "Decreased monitoring visit cycle time by 22% by shifting to risk-based monitoring and improving issue triage in Medidata Rave." |
| Audit readiness | "Achieved zero critical findings in a sponsor audit by closing 95% of action items within 10 business days using a CAPA tracker." |
Turn your everyday tasks into measurable, recruiter-ready resume bullets in seconds with Enhancv's Bullet Point Generator.
With strong bullet points in place, the next step is ensuring your skills section presents the right mix of hard and soft skills that clinical trial management demands.
How to list your hard and soft skills on a clinical trial manager resume
Your skills section shows you can run compliant, on-time trials, and recruiters and ATS scan this section to confirm you match the protocol, vendor, and regulatory requirements; aim for a skills mix weighted toward hard skills with role-specific soft skills.
clinical trial manager roles require a blend of:
- Product strategy and discovery skills
- Data, analytics, and experimentation skills
- Delivery, execution, and go-to-market discipline
- Soft skills
Your skills section should be:
- Scannable (bullet-style grouping).
- Relevant to the job post.
- Backed by proof in experience bullets.
- Updated with current tools.
Place your skills section:
- Above experience if you're junior or switching careers.
- Below experience if you're mid/senior with strong achievements.
Hard skills
- ICH E6 GCP compliance
- Study start-up, site activation
- Protocol, ICF development
- Clinical trial budgeting, forecasting
- Vendor management, CRO oversight
- Risk-based monitoring, CAPA
- TMF management, eTMF systems
- CTMS administration, reporting
- EDC systems, data cleaning
- Safety reporting, SAE workflows
- Regulatory submissions, IRB/IEC
- Quality management systems (QMS)
Soft skills
- Lead cross-functional trial teams
- Align stakeholders on timelines
- Escalate risks with options
- Make trade-off decisions fast
- Run sponsor and investigator calls
- Negotiate scope with vendors
- Drive issue resolution to closure
- Communicate protocol changes clearly
- Maintain audit-ready documentation habits
- Coach coordinators and monitors
- Manage competing priorities calmly
- Hold teams accountable to milestones
How to show your clinical trial manager skills in context
Skills shouldn't live only in a bulleted list on your resume. Explore resume skills examples to see how top candidates integrate competencies throughout their documents.
They should be demonstrated in:
- Your summary (high-level professional identity)
- Your experience (proof through outcomes)
Here's what strong, skills-rich content looks like in practice.
Summary example
Clinical trial manager with 12 years in oncology research, skilled in Medidata Rave, risk-based monitoring, and cross-functional vendor oversight. Led 15+ Phase II–III trials, reducing protocol deviations by 34% through proactive site engagement.
- Reflects senior-level expertise clearly
- Names industry-standard tools directly
- Quantifies impact with real metrics
- Highlights leadership and collaboration
Experience example
Senior Clinical Trial Manager
Veridian Therapeutics | Remote
March 2019–August 2024
- Managed five concurrent Phase III oncology trials across 40 global sites using Medidata Rave, achieving 97% data query resolution within 48 hours.
- Partnered with regulatory, biostatistics, and CRO teams to streamline monitoring plans, cutting site activation timelines by 22%.
- Implemented risk-based monitoring frameworks that reduced protocol deviations by 31% across two pivotal registration studies.
- Every bullet includes measurable proof.
- Skills surface naturally through outcomes.
Once you’ve demonstrated your clinical trial manager capabilities through specific, results-focused examples, the next step is adapting that approach for a resume when you don’t have direct experience.
How do I write a clinical trial manager resume with no experience
Even without full-time experience, you can demonstrate readiness through clinical research and related work. Writing a resume without work experience requires focusing on transferable skills and project-based accomplishments:
- Clinical research coordinator internships.
- Academic clinical trial capstone projects.
- Site startup and activation support.
- Regulatory document and binder work.
- Electronic data capture sandbox builds.
- Institutional review board submissions support.
- Good Clinical Practice certification training.
- Quality management system audits participation.
Focus on:
- Trial lifecycle ownership examples.
- Regulatory and compliance deliverables.
- Metrics, timelines, and budgets.
- Tools: CTMS, EDC, eTMF.
Resume format tip for entry-level clinical trial manager
Use a hybrid resume format because it highlights trial-relevant projects and tools while still showing steady education and work history. Do:
- Lead with a summary naming clinical trial manager targets.
- Add a projects section with trial scope.
- List tools: CTMS, EDC, eTMF.
- Quantify deliverables, timelines, and error rates.
- Mirror job posting keywords in bullets.
- Built an EDC sandbox in REDCap for a five-visit study, created edit checks and queries, and reduced missing data from twelve percent to four percent.
Even without direct experience, your educational background can serve as one of the strongest foundations for your clinical trial manager resume—so presenting it effectively matters.
How to list your education on a clinical trial manager resume
Your education section helps hiring teams confirm you hold the scientific or clinical foundation this role demands. It validates your readiness to manage complex trial operations.
Include:
- Degree name
- Institution
- Location
- Graduation year
- Relevant coursework (for juniors or entry-level candidates)
- Honors & GPA (if 3.5 or higher)
Skip month and day details—list the graduation year only.
Here's a strong education entry tailored to the clinical trial manager role.
Example education entry
Bachelor of Science in Clinical Research
University of North Carolina at Chapel Hill, Chapel Hill, NC
Graduated 2019
GPA: 3.7/4.0
- Relevant coursework: Biostatistics, Good Clinical Practice, Regulatory Affairs, Pharmacology, and Research Ethics
- Honors: Magna Cum Laude, Dean's List (all semesters)
How to list your certifications on a clinical trial manager resume
Certifications show your commitment to continuous learning, proficiency with clinical research tools, and relevance to regulated trial environments as a clinical trial manager.
Include:
- Certificate name
- Issuing organization
- Year
- Optional: credential ID or URL
- List certifications below education when they're older, less role-specific, or secondary to your degree and core clinical trial manager experience.
- List certifications above education when they're recent, highly relevant, or required for the clinical trial manager roles you target.
Best certifications for your clinical trial manager resume
- Certified Clinical Research Professional (CCRP)
- Certified Clinical Research Associate (CCRA)
- Clinical Research Coordinator (CRC) Certification
- Certified Project Manager (CPM)
- Project Management Professional (PMP)
- Good Clinical Practice (GCP) Certification
- Regulatory Affairs Certification (RAC)
Once you’ve positioned your credentials where recruiters can find them, use your clinical trial manager resume summary to connect those qualifications to the value you bring at a glance.
How to write your clinical trial manager resume summary
Your resume summary is the first thing a recruiter reads, so it must immediately signal your fit for the role. A strong opening sets the tone and determines whether the rest of your resume gets attention.
Keep it to three to four lines, with:
- Your title and total years of clinical trial management experience.
- Therapeutic areas, phases, or trial types you specialize in.
- Core tools and skills such as CTMS, EDC systems, ICH-GCP, and regulatory submissions.
- One or two measurable achievements like enrollment targets met or cycle times reduced.
- Soft skills tied to outcomes, such as cross-functional leadership that improved site performance.
PRO TIP
At this level, focus on your ability to lead complex trials, manage CROs, and drive timelines. Emphasize decision-making authority, budget oversight, and regulatory outcomes. Avoid vague descriptors like "motivated" or "passionate." Replace them with specific accomplishments that show operational impact and strategic value.
Example summary for a clinical trial manager
Clinical trial manager with eight years of experience leading Phase II–III oncology trials across 40+ global sites. Reduced enrollment timelines by 22% through CRO oversight and proactive risk management.
Optimize your resume summary and objective for ATS
Drop your resume here or choose a file.
PDF & DOCX only. Max 2MB file size.
Now that your summary conveys your qualifications at a glance, make sure your header provides the essential contact details recruiters need to actually reach you.
What to include in a clinical trial manager resume header
A resume header lists your key identifiers and contact details, boosting visibility, credibility, and recruiter screening for a clinical trial manager role.
Essential resume header elements
- Full name
- Tailored job title and headline
- Location
- Phone number
- Professional email
- GitHub link
- Portfolio link
A LinkedIn link helps recruiters verify your experience quickly and supports screening.
Do not include photos on a clinical trial manager resume unless the role is explicitly front-facing or appearance-dependent.
Keep the header to two lines, match the clinical trial manager job title to the posting, and use consistent formatting across all links.
Example
Clinical trial manager resume header
Jordan Lee
Clinical Trial Manager | Phase II–III Operations, Site Activation, and Vendor Oversight
Boston, MA | (617) 555-01XX | jordan.lee@enhancv.com | github.com/jordanlee | jordanlee.com | linkedin.com/in/jordanlee
Once your contact details and role-specific identifiers are in place, add targeted additional sections to strengthen your clinical trial manager resume and support the information above.
Additional sections for clinical trial manager resumes
When your core qualifications match other candidates, additional sections can set you apart and reinforce your credibility as a clinical trial manager.
- Languages
- Publications and research contributions
- Professional affiliations (e.g., ACRP, SOCRA, DIA)
- Conferences and speaking engagements
- Volunteer experience in clinical research
- Certifications and continuing education
Once you've rounded out your resume with sections that highlight your full professional profile, it's worth pairing it with a cover letter to add even more context to your application.
Do clinical trial manager resumes need a cover letter
A cover letter isn't required for a clinical trial manager, but it often helps. Understanding what a cover letter is and when to use one can matter most in competitive roles, sponsor-facing teams, or when hiring managers expect a clear narrative beyond the resume.
Use a cover letter to add context that a resume can't:
- Explain role and team fit: Align your experience with the trial phase, therapeutic area, vendor model, and cross-functional partners.
- Highlight one or two outcomes: Name a study milestone you improved, such as faster site activation, fewer protocol deviations, or cleaner data.
- Show business and user understanding: Connect your work to patients, investigators, sponsors, timelines, budgets, and inspection readiness.
- Address transitions or non-obvious experience: Clarify a move from coordinator to clinical trial manager, a therapeutic area switch, or a gap with relevant skills.
Drop your resume here or choose a file.
PDF & DOCX only. Max 2MB file size.
Even if you choose not to include a cover letter, using AI to improve your clinical trial manager resume helps you strengthen the document that carries the most weight in your application.
Using AI to improve your clinical trial manager resume
AI can sharpen your resume's clarity, structure, and impact. It helps tighten language and highlight results that matter to hiring managers. But overuse creates generic, hollow content. If you're wondering which AI is best for writing resumes, the answer depends on your specific needs—but once your resume feels clear and aligned with your target role, step away from AI.
Here are 10 prompts you can copy and paste to strengthen specific sections of your clinical trial manager resume:
- Sharpen your summary: "Rewrite my clinical trial manager resume summary to emphasize leadership, regulatory expertise, and measurable outcomes in under four sentences."
- Quantify experience bullets: "Add specific metrics to these clinical trial manager experience bullets, focusing on enrollment targets, timelines, and budget management."
- Align skills strategically: "Compare my skills section against this clinical trial manager job description and suggest missing hard skills I should add."
- Strengthen action verbs: "Replace weak or repetitive verbs in my clinical trial manager experience section with precise, high-impact alternatives."
- Tighten project descriptions: "Edit my clinical trial manager project entries to clearly state study phase, therapeutic area, team size, and key outcomes."
- Refine education entries: "Rewrite my education section to highlight coursework and research relevant to a clinical trial manager role."
- Optimize certification placement: "Suggest the best way to present my ACRP and SOCRA certifications on a clinical trial manager resume."
- Eliminate redundancy: "Identify and remove redundant phrases across all sections of my clinical trial manager resume without losing key details."
- Tailor for ATS: "Restructure my clinical trial manager experience bullets to naturally incorporate these keywords from the job posting."
- Clarify compliance impact: "Rewrite these bullets to clearly show how my clinical trial manager work ensured FDA or ICH-GCP compliance."
Stop using AI once your resume sounds accurate, specific, and aligned with real experience. AI should never invent experience or inflate claims—if it didn't happen, it doesn't belong here.
Conclusion
A strong clinical trial manager resume proves impact with measurable outcomes, role-specific skills, and a clear structure. It highlights enrollment performance, timeline delivery, budget control, and audit readiness. It shows expertise in protocol execution, vendor oversight, risk management, and cross-functional leadership.
Keep each section easy to scan and focused on results, scope, and tools. This approach matches today’s hiring market and supports near-future expectations for speed, compliance, and data quality. A well-built clinical trial manager resume helps recruiters assess fit fast.